Ideas in Medicine bring novel product to market
Approach
Gastric Light Guide Inc, lacking any commercial experience, approached Ideas in Medicine to help bring this novel product to market. Impressed with the technology and the tremendous benefits it could bring to doctors, patients and the healthcare system, GL Inc and IIM decided the best option was for IIM to assume responsibility for further product development, marketing, regulatory approvals, IP protection, manufacturing and global distribution. The genesis of Ideas in Medicine was to service the gap between idea and commercial product. In Dr Dib’s case he is a busy surgeon with neither the time nor experience to bridge that gap. Ideas in Medicine is uniquely qualified and ideally suited for brilliant minds with great product ideas that lack a go-to-market strategy.
Results
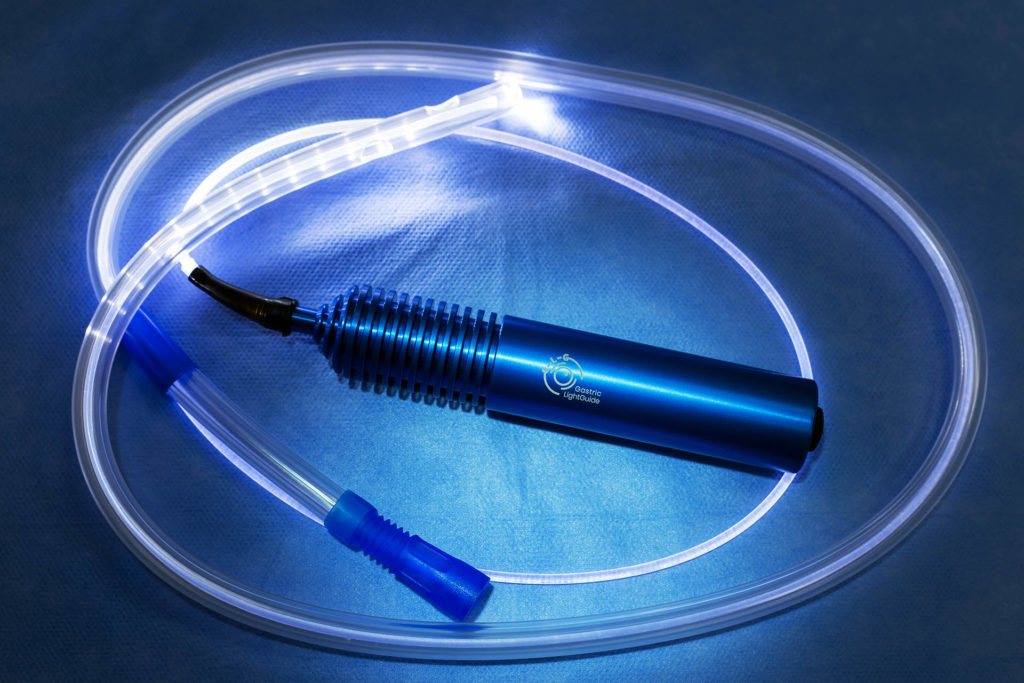
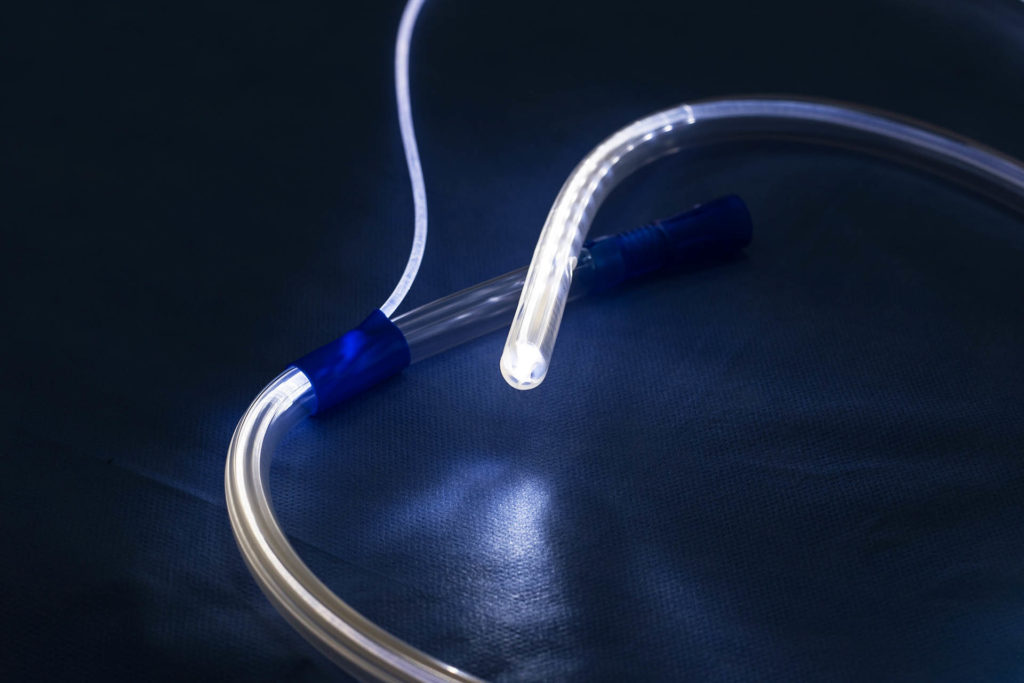
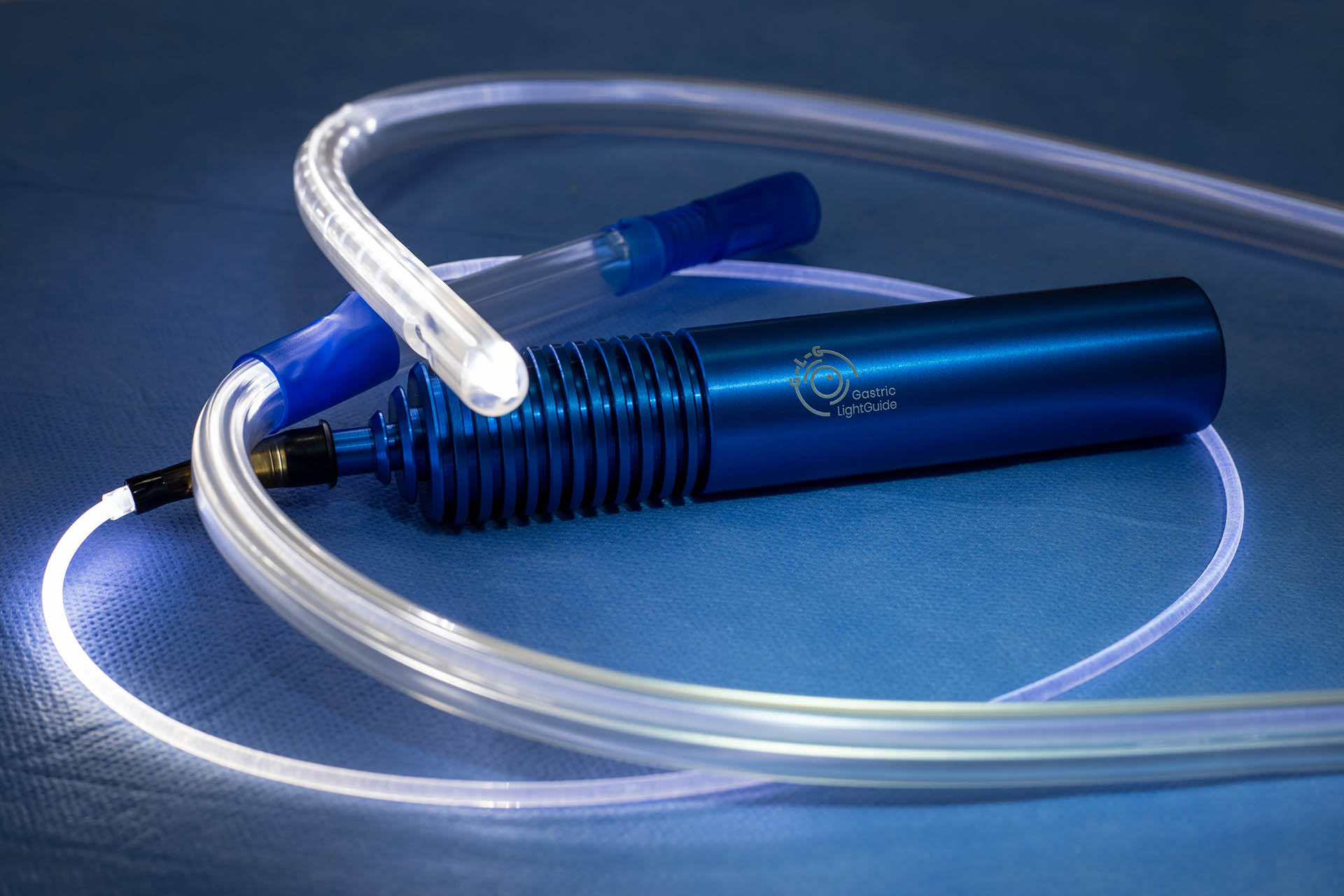
Ideas in Medicine’s input expedited the Gastric Light “design freeze” by proposing and integrating several key features that enhanced the user experience as well increased the saleability of the product. IIM created the final versions of product packaging and labeling, added key language to IP filings to expand protection, initiated FDA clearance, created all marketing collateral, and created distribution channels in the EMEA, Asia and South America. Once FDA clearance is obtained a US launch plan will be finalized. Using industry press contacts IIM facilitated an article on the Gastric Light Guide, which appeared in Bariatric News (distributed globally) as seen in the link below.
https://bariatricnews.net/?q=news/113672/gastric-light-illuminating-sleeve-gastrectomy-procedures